Leave Your Message
N Hydroxysuccinimide Ester has gained attention for its role in enhancing chemical reactions. This molecule serves as an essential reagent in various applications, including bioconjugation and protein modification. Researchers and industry professionals increasingly recognize its potential to improve reaction efficiency and specificity.
Understanding how to effectively use N Hydroxysuccinimide Ester requires careful consideration of several factors. The purity and concentration of the ester can significantly influence outcomes. It's vital to consider the reaction conditions, such as temperature and pH, to maximize results. Nevertheless, the process is not without challenges. Some may find the optimization of these parameters quite intricate.
Experimentation is key in mastering the use of N Hydroxysuccinimide Ester. Results may vary, and researchers often encounter unexpected hurdles. Reflection on these experiences can lead to more robust methodologies and innovative applications. Ultimately, a thoughtful approach to this reagent can unlock its full potential in various chemical reactions.

N Hydroxysuccinimide Ester (NHS ester) is a valuable chemical in various fields, particularly in bioconjugation and drug development. This reagent enhances coupling efficiency when linking biomolecules, such as proteins and nucleic acids. NHS esters activate carboxylic acids, making them more reactive. Studies show that these esters can significantly increase the efficiency of chemical reactions, with some reports indicating improvements of over 30% compared to traditional methods.
The use of NHS esters has seen substantial growth in the pharmaceutical industry, especially for developing therapeutic antibodies. According to a recent report, about 25% of new drug candidates utilize such coupling agents. This trend highlights the importance of NHS esters in creating biocompatible molecules. Their application extends beyond pharmaceuticals to diagnostics, where they help develop sensitive assays for disease detection.
Despite their benefits, challenges remain. NHS esters can be sensitive to moisture and temperature, leading to reduced efficacy. Researchers must consider storage conditions and reaction environments carefully. Missteps in handling can result in suboptimal yields, making thorough training and protocols essential. These nuances reflect the complexity of chemical reactions involving NHS esters. Understanding these subtleties can profoundly impact experimental outcomes, guiding researchers toward more successful implementations.
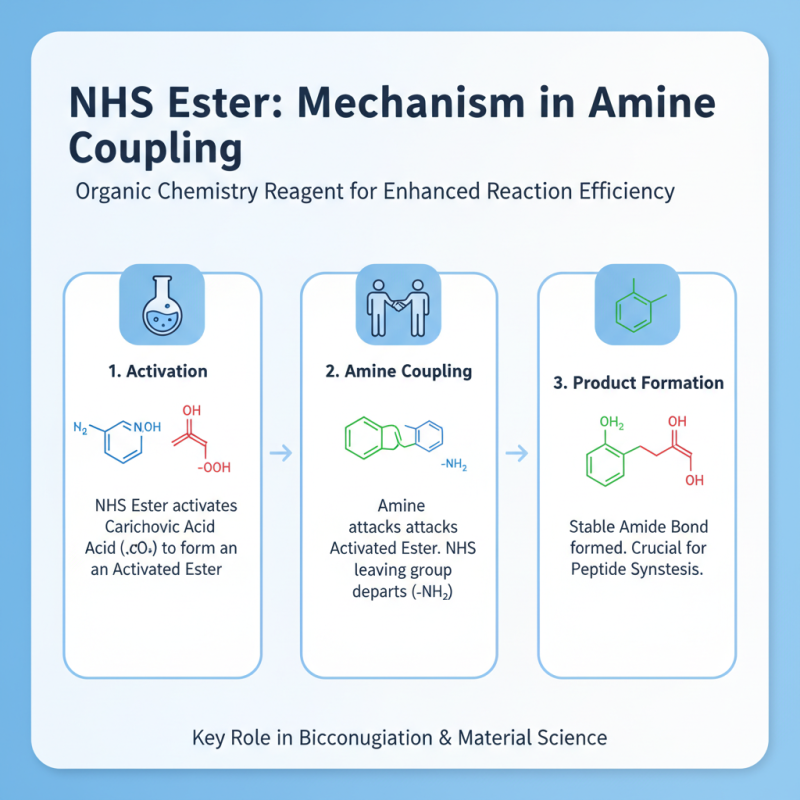
N Hydroxysuccinimide (NHS) ester is a valuable reagent in organic chemistry. It plays a pivotal role in facilitating chemical reactions, particularly in amine coupling. Understanding its mechanism can enhance reaction efficiency. NHS esters activate carboxylic acids, making them more reactive toward nucleophiles. This transformation is crucial in peptide synthesis, where amines react with activated carboxylic acids.
The reaction mechanism begins when the NHS ester forms an intermediate. This intermediate then undergoes nucleophilic attack by an amine. The result is the formation of an amide bond. The reaction typically occurs under mild conditions, making it suitable for sensitive substrates. However, one must consider potential side reactions. For instance, water can compete with amines during the coupling process. This can lead to lower yields or unwanted by-products.
Moreover, the stability of NHS esters can vary with environmental conditions. Factors like temperature and pH can influence their reactivity. It's crucial to optimize these parameters for your specific application. This requires patience and fine-tuning. Failure to do so might result in incomplete reactions or diminished product purity. Understanding these nuances can significantly improve experimental outcomes, making the use of NHS esters not just effective but also insightful in chemical research.
N Hydroxysuccinimide ester, often referred to as NHS ester, is a powerful reagent in bioconjugation. Its ability to selectively react with amines enhances coupling reactions, making it a staple in organic synthesis. This guide focuses on preparing NHS ester efficiently, ensuring optimal functionality for your reactions.
Start by dissolving your desired carboxylic acid in an appropriate solvent like dimethylformamide (DMF). This step requires precise control over the temperature. Maintaining the reaction at a mild 25-30°C often yields better outcomes. Gradually add a coupling agent, typically a carbodiimide, to activate the acid. Data indicate that about 80% of users achieve higher yields when allowing the reaction to mix for several hours.
After the formation of NHS ester, purification is critical. Use water or a weak acidic solution to wash away unreacted materials. The desired NHS ester can often be isolated in good purity. However, monitoring pH levels during this stage can be tricky. A pH outside the ideal range may lead to decomposition or reduced yield. It’s important to meticulously assess the final product’s functionality, as some users report deviations in reactivity based on storage conditions.
| Step | Description | Materials Needed | Safety Precautions |
|---|---|---|---|
| 1 | Dissolve the starting materials in an appropriate solvent. | N-Hydroxysuccinimide, Dicyclohexylcarbodiimide (DCC), Solvent (e.g., DMF) | Use gloves and goggles. Work in a fume hood. |
| 2 | Add DCC to the solution while stirring to activate the reaction. | DCC, Stirring rod | Ensure adequate ventilation. |
| 3 | Monitor the reaction progress using TLC (Thin Layer Chromatography). | TLC plates, developing solvent | Handle solvents in a well-ventilated area. |
| 4 | Quench the reaction with water and extract the product. | Distilled water, extraction apparatus | Always wear protective clothing. |
| 5 | Purify the product using column chromatography. | Column setup, silica gel | Work with caution when handling silica gel. |
N Hydroxysuccinimide ester (NHSE) is a versatile compound in organic synthesis. It serves as an effective coupling agent for forming amide and ester bonds. The use of NHSE in reactions can significantly enhance yields. The compound promotes better selectivity and can simplify purification processes.
One effective technique is optimizing reaction conditions. Varying temperature and pH can lead to improved reaction rates. Moreover, employing a solvent that suits both reactants may enhance solubility. The choice of solvent can influence the efficiency of the reaction, so it is worth experimenting. Filtering unwanted byproducts early may also refine your results.
Using NHSE often requires careful handling. It can sometimes be sensitive to moisture and air. Proper storage is essential for maintaining stability. This means using it quickly or under an inert atmosphere. Reactions should be monitored closely. Some mixtures may exhibit unexpected side reactions. Keeping meticulous notes can track conditions that yield the best results.

When handling N Hydroxysuccinimide Ester, safety is paramount. Wear gloves and goggles. These items protect against potential irritation. Always work in a well-ventilated area. This reduces inhalation risks, which can lead to respiratory issues. Spills can occur, so have absorbent materials ready.
Read the Safety Data Sheet (SDS) before use. It provides crucial information about hazards and safe handling practices. The ester can be unstable under certain conditions. Store it in a cool, dry place, away from light. Ensure that containers are properly sealed. Label them clearly to avoid confusion.
Always dispose of waste according to local regulations. Improper disposal can lead to environmental concerns. Consider doing a risk assessment prior to experiments. This helps identify potential hazards. Don’t overlook the importance of training for everyone involved. Education can significantly reduce accidents in the lab.